The Good, the Bad and the Ugly: a review of SARS Lab Escapes
Research by DRASTIC, a group of researchers working together to investigate the origins of SARS-CoV-2, starting from facts and not bending to any pressure. DRASTIC members were the first ones to discover the Mojiang ‘miners’ story.
DOI: 10.5281/zenodo.4293257
Introduction
In 2003–04, in the wake of the SARS epidemics, there were multiple cases of laboratory acquired infection (LAI) with SARS in just a few months: first in a P3 in Singapore, then in a P3 lab that was part of a military P4 complex in Taipei, and last a protracted case in a P3 in Beijing. The ‘WHO SARS Risk Assessment and Preparedness Framework’ has a good summary of these lab accidents:
Since July 2003, there have been four occasions when SARS has reappeared. Three of these incidents [note: Singapore, Taipei and Beijing] were attributed to breaches in laboratory biosafety and resulted in one or more cases of SARS. The most recent laboratory incident [note: in Beijing] resulted in 9 cases, 7 of which were associated with one chain of transmission and with hospital spread. Two additional cases at the same laboratory with a history of illness compatible with SARS in February 2004 were detected as part of a survey of contacts at the facility.
[i.1]
This article reviews some of these cases and discusses briefly some of the insights that were gained from these at the time.
1. The Good: Singapore P3 — Sep 2003
In a few words: This first SARS LAI accident (in Singapore) exposed some serious structural biosafety issues, but the investigation was very thorough and transparent, and even extended into a review of all BSL-3s in the city-state. The Singapore government used it as an opportunity to fundamentally redesign its biosafety approach, just at the time when more BSL-3s were due to come in line. We also note that the (strongly controlled) local media do not seem to have contributed any additional details to the story.
1.1 The Accident:
In September 2003, a 27-year-old student from the National University of Singapore (NUS) was found to be infected with the SARS virus due to improper experimental procedures. [s.1]
The student was in his third year of a doctoral program in microbiology and was studying the replication of flaviviruses such as the West Nile Virus (which was just causing a peak of infection in the US at the time) in cells using electron microscopy. So far, he had worked at the NUS BSL- 2 with the attenuated Sarafend strain of the West Nile virus. Because he wanted to compare this attenuated train with the more pathogenic New York strain of the virus (that one a BSL-3 pathogen), and since NUS had no BSL-3, an agreement was made for him to do this work at the BSL-3 facility of Institute of the Environmental Health Institute (EHI) of Singapore.
The BSL-3 of the EHI had been heavily involved in SARS research during the recent outbreak and was continuing that work at the time [s.6].
The student had no previous experience working in a BSL-3. On his first visit to the EHI, he was given only 20 minutes training with BSL-3 procedures before entering the BSL-3 lab.
On his second and last visit to the institute (Saturday 23 Aug 2003) he entered the lab 3 times:
- The first time, he went in with the technician, wearing only street clothes, and did not engage in any work.
- The second time, staff of the EHI were meeting. Before going to that staff meeting, the virology technician had grown up a stock of the New York isolate and centrifuged the supernatant from infected cells. The technician had then placed the centrifuge tubes in the Class II biological safety cabinet for the student to ampoule as a seed stock for his research. Accordingly, the student put on a gown and two sets of gloves before entering the laboratory alone, where he spent 20 minutes unsupervised, transferring the cell supernatant into pre-labelled cryovials under the Class II biosafety hood. [s.6]
- The third time he re-entered the lab with the technician who was back from her meeting and transferred the cryovials to a –70°C freezer located in the BSL-2 facility, as no such freezer existed inside the BSL-3 laboratory, so that all frozen BSL-3 virus strains were effectively stored in the BSL-2 freezer.
The student did not engage in any more work with these before falling ill three days later on Aug 26, developing mild SARS symptoms. He sought outpatient medical care from his general practitioner, Singapore General Hospital (SGH) emergency room and a Chinese physician. He eventually returned to the SGH with persistent fevers and was admitted to the hospital on Sep 3. Fortunately, he recovered and there were no secondary cases.
1.2 The Investigation:
Analysis of the ampoules of seed West Nile virus that the student stored on that 23rd Aug showed that the vials contained SARS-CoV as well as West Nile virus. That SARS-CoV matched the SARS isolate that was handled at the EHI.
Poor record keeping made it difficult to ascertain if there was a live SARS virus in the BSL-3 laboratory on that exact 23rd Aug, but it was established that there was some there 2 days before. [s.8]
In other words there had been a SARS-contamination within the BSL-3.
A 11-member review panel led by Antony Della-Porta, Biosafety Expert for the WHO [t.3], produced a report for the Ministry of Health in Singapore. The panel concluded that a combination of
“Inappropriate laboratory standards and a cross-contamination of West Nile virus samples with SARS coronavirus in the laboratory led to the infection of the doctoral student”.
[s.8]
The investigators also documented a variety of shortcomings within the EHI BSL-3 lab that most likely contributed to the incident. These included inadequate record-keeping procedures, totally inadequate training, inexistent virus stock inventory, patchy maintenance records plus a variety of structural problems including the absence of gauges to indicate the pressure differentials, the lack of a freezer to store samples, problems with HEPA filters and air supply, and other equipment deficiencies. [s.5, s.8].
Accordingly, they first recommended that all BSL-3 work cease there ‘until the laboratory deficiencies have been addressed and subjected to external audit’. [s.8]
Note: While many circumstantial evidences pointed to a real risk of SARS contamination of the EHI BSL-3 lab, the exact causes of this particular accident — such as a precise bad manipulation or the failure of a precise piece of equipment — was never precisely identified.
1.3 Structural Issues:
At the invitation of the Singapore Ministry of Health, the investigation team went further than the EHI P3 lab and looked also at overall biosafety practices in other Singapore institutions. This was both a very courageous and very useful initiative, especially in view of the large number of new Singapore BSL-3 labs that were supposed to come in line soon at the time.
The Singapore General Hospital BSL-3, the NUS BSL-2 and the Defence Science Organisation were reviewed. The panel found deficiencies at the Singapore General Hospital as well as at the NUS BSL-2. [s.8] and made recommendations to improve biosecurity in each institution they visited.
More generally, the panel concluded that there were no appropriate standards, regulations, or guidelines on biosafety in Singapore, and offered some recommendations [s.8] as to what could be done to rectify the situation:
Biological Standards
Recommendation 7.1: Need for a National Legislative basis for Standards in Biosafety Laboratories for Singapore.[..].
Recommendation 7.2: A structure should be created for laboratory certification covering both structure integrity and operating procedures. [..]
Recommendation 7.3: Creation of the tracking system for importation, exportation to and from Singapore. [..]
1.4 The Aftermath:
At the end of September, Lim Ruisheng, Minister of Environment of Singapore, apologised to the people of Singapore for the SARS infection incident in the laboratory of the Institute of Environmental Health. Lin Ruisheng said:
“The Environmental Health Research Institute must take responsibility, and the National Environmental Administration must also take responsibility. As the Minister of Environment, I should be more responsible. Because the investigation results of the investigation team show that our laboratory is indeed not safe enough.”
[s.3]
Since 2003, Singapore has introduced legislation and regulations to cover the handling of high risk infectious agents and developed a laboratory accreditation system. Aligning incentives with regulation and training, they have also taken a number of initiatives to reward outstanding safety practices.[s.6]
2. The Bad: Taiwan Military P4 — December 2003
In a few words: The second SARS LAI accident (Taiwan) was a less straightforward affair. First, the human factor played a big role in delaying notification to the authorities. Then the accident had a large epidemic potential as it involved international air-travelling shortly after infection. Furthermore, while the official reporting of the accident was generally transparent, a few aspects of the story remained somewhat opaque due to the military setting.
Taiwanese media, and interestingly also mainland China media, have contributed circumstantial details to the story. Generally, the reaction of the authority seems to have been earnest and constructive.
2. 1 The Accident:
Oddly, most references to this lab accident simply fail to mention that it happened in a military P4 laboratory (likely built or equipped by France) utilising type III glove ported isolators [t.17]. These facts are particularly important and should be better publicised.
This lab escape involved the Taiwan Military Institute of Preventive Medical Research (IPMR) of the National Defence University. It is located in Baiji Mountain, Sanxia (or Sanhsia, 三峽, Three Georges), on the outskirts of Taipei Town, and is allegedly partly dug into the hill. The laboratory is known as the centre of Taiwan’s biochemical capacities. The facility was allegedly capable of producing weapons-grade biological agents [t.1] and may have done so in the past [t.18], although Taiwanese officials claim it exists only to research ways of preventing biological attacks from mainland China.
According to unverified Chinese sources, Taiwan purchased the IPMR from France in 1983 [t.2]. A more recent Western source described it as being ‘full of advanced French-made equipment’ [t.18]
As the top biosafety lab in Taiwan, the IPMR has been racing against the clock to analyse the characteristics of the virus since the SARS outbreak. In May 2003, President Chen Shui-Bian had announced that he had ordered the IPMR to work on a vaccine for SARS. [t.18, t.20]
On December 6th, 2003, 44-year-old Lieutenant-Colonel Chan Jiacong (危家琼), a Ph.D. in pathology from Johns Hopkins University (US), contracted SARS at the IPMR due to negligence.
The researcher worked with SARS. He was cleaning his Taipei lab when he
found a ripped bag [t.7] in the negative-pressure transport cabinet of the laboratory. He assumed that the bag had been leaking for several days and may have thought the virus had already lost its effectiveness. In any case, the researcher was working alone and had to travel to Singapore the next day to address a meeting about his SARS research, so he was in a rush t.4, t.10]. [Note: we were unable to identify a suitable conference in Singapore on those days — suggestions welcome]
The proper way of cleaning that spill was to use vaporised hydrogen peroxide, but this would have taken hours. Instead, he decided to use a shortcut in the form of 70% ethanol, which he could spray on the spill before wiping it out [t.17].
As he could not reach the spot with the gloves attached to the cabinet, he instead wore a “normal mask” and surgical gloves, no protective gown, opened the transport cabinet, tried to reach the spill — but because he still could not reach it, he then put his head inside the cabinet pass-through to spray the spill with the ethanol mix. After spraying it and waiting for 10 minutes, he wiped the spill out.
In the process it seems that he then simply dumped the leaking bag on his trash cart.[t.2, t.7, t.8, t17].
“He was in a hurry to get ready for a conference in Singapore, so he was rushing to finish his disinfection work and was careless”
Su Ih-jen, chief of Taiwan’s Centre for Disease Control [t.5]
Another source notes that:
[The] laboratory was also found to have violated many safety and record-keeping standards. For example, this researcher regularly worked long shifts (12 to 14 hours) usually alone and there was no timely procedure in place for reporting incidents. In addition, there was no record of him actually working in the laboratory since he had recently lost his building access card and was using a borrowed card on the date of the incident.
Della-Porta explains that the recommended procedure for decontamination of the safety cabinets used in the Taiwan facility is the use of a hydrogen peroxide generator, which takes several hours. He adds that a shortcut of 70 percent ethanol for 10 minutes is completely inadequate. In addition, there were inadequate standard operating procedures and Taiwan was without guidelines or regulations related to biological safety.’
[t.10]
On the following day, Dec. 7, he flew to Singapore with six laboratory colleagues on a China Airlines flight. He complained of fatigue and muscle pain on Dec. 8 while in Singapore, but had no fever until the night of Dec. 10 after he returned to Taipei. He later had diarrhoea.
Soon after returning from Singapore, he reported to the Defence University clinic that he had developed influenza. Among many issues, the WHO investigation team recorded a failure to have a medical monitoring programme in place. Indeed, senior management could not explain why, after Chan had reported to the Defence University clinic with a respiratory disease, he was not followed up for the next 6 days when he was absent from work. [t.16]
2.2 Fear of Losing face and bringing shame:
Faced with the standard symptoms of SARS, Lieutenant-Colonel Chan was very aware that he may have caught SARS. However, he decided to self-isolate at home, relying on his father to care for him, as he was unwilling to seek medical care because he dreaded bringing disgrace to himself and his institution.
His father eventually persuaded him [t.6] to seek medical attention by threatening to commit suicide. He checked into Taiwan Hoping Hospital only on Dec 16 by which time he had developed other SARS symptoms, such as a cough and signs of pneumonia. He was readily diagnosed as having SARS [t.16].
His father said in a television interview that his son knew early on that he might have SARS. But he delayed going to the hospital because he didn’t want to panic the public and disgrace Taiwan. He quarantined himself at home until Dec. 16 and relied on his father for food and other care.
His father told CTI cable TV that his son: “wanted to die at home because he feared his illness would bring shame to his lab and the country”.
The father said he pleaded with his son to seek treatment. As the scientist’s condition worsened, his father said he threatened to commit suicide if he didn’t get help. “He finally agreed to go to the hospital after I threatened to kill myself,” his father said. “He was thinking of the country. He’s a military man, and military men are bound by a natural duty,”
[t.6]
2.3 Reaction from the Authorities
A chest x-ray showed pneumonia in his right lung and polymerase chain reaction tests of throat and blood samples were positive for the SARS virus. The finding was further confirmed on multiple samples in two laboratories in Taipei.
Six colleagues were ordered to suspend work at the laboratory and start self-isolation at home for 21 days. The four family members of the officer self-isolated at home for 21 days, although they had not displayed fever or any other SARS-like symptoms.
Although Chan was not in the contagious phase when he travelled, around 95 people who had been in contact with him were quarantined until Dec. 23, including 19 that were on his flights and 79 in Singapore. None of them were infected. It is worth noting that officials were having difficulty contacting three Americans, a Japanese, and a Singaporean who had been on the flight. Additionally, two of his colleagues went to Singapore with the researcher on Dec. 7 and later travelled to the United States. [t.6, t.14]
Five foreigners — three Americans, one Japanese and a Singaporean — who sat near him on the return flight from Singapore have been located and told to monitor their health, officials said on Friday. The Japanese traveler was located in Taiwan where he lives, two of the Americans went to Hong Kong and the other is in the United States, said Shih Wen-yi, a spokesman for Taiwan’s Center for Disease Control. They were told to be on alert for SARS symptoms, like a fever.
[t.6]
The Taiwan Science Council closed the IPMR temporarily and issued a resolution stating that Lieutenant Colonel Chan violated the “Laboratory Safety Guidelines and Norms” of the SARS project research plan and was given a sanction not to apply for research project funding.
The laboratory conducting severe acute respiratory syndrome (SARS) research at the Institute of Preventive Medicine Research (IPMR) of the National Defence Medical College was closed down temporarily Wednesday after one of its researchers was confirmed to have contracted the flu-like disease, military sources said.
[t.6]“All SARS research work in the military has also been halted”
Chang Sheng-yuan, director-general of Taiwan’s Military Medicine Bureau
The CDC conducted two complete environmental disinfections. All equipment was inspected in detail, and all personnel were retrained. [t.2, t.13]
Separately, in China, authorities ordered all researchers to hand in SARS samples as a precaution. China’s Health Ministry told laboratories to send samples to “designated places for storage” and “demanded that all regions strengthen management of the SARS virus,” the official Xinhua News Agency said. [t.6]
2.4 WHO enquiry and international cooperation:
A WHO Team was invited to investigate the case. Anthony Della-Porta, who had earlier headed the WHO investigation of the SARS LAI assisted with the investigation.
As far as we can tell, the WHO investigation seems to have been rather comprehensive and transparent, finding and reporting quite a few failures, with the exact circumstances of the accident well delineated [t.13]. The international cooperation effort that followed seems to have been constructive and certainly helped rebuild trust in the Taiwanese capacity to manage their BSL3/4s:
In January 2004, the World Health Organization (WHO) dispatched an expert team [of 3] led by Dr. Antony Della-Porta to Taiwan because of the SARS case (Figure 1). They visited five existing and new constructions of BSL-3 laboratories during their short stay. In addition to the advice given to those individual laboratories, four major measures in terms of general management of biological safety were suggested.
Soon after, CDC invited two world-class experts in the field, Dr. Thomas Ksiazek from U.S. CDC and Dr. Kazuyoshi Sugiyama from National Institute of Infectious Diseases (NIID) of Japan, respectively, in March and November 2004, to give [Taiwan CDC] us a helping hand in inspecting current safety and management of equipment in laboratories of Biosafety Level 3 or above across Taiwan (Figure 2).
[t.9]

As the Taiwan CDC would later state,
‘The laboratory-acquired case of SARS in Taiwan sent a shockwave through the nation. However, this incident was a crucial turning point. It led to the seeking and adoption of advice from both national and international experts and scholars and, therefore, acquired invaluable learning experience in biological safety management’.
[t.11]
2.5 Lessons to be learnt:
Henk Bekedam, the WHO representative in China, told reporters in Beijing that the case in Taiwan should alert scientists:
“It’s a clear reminder again that we have to be extremely cautious working with the SARS coronavirus and there are whole issues about whoever in the whole world is keeping a SARS specimen has to be very careful in dealing with this”
[t.6]
Anthony Della-Porta was even more clear:
“It hasn’t been a disaster, but it could have been.”
[t.12]
As noted by Furmanski [t.15] (quoted here), this second outbreak further shook the virology communities in Asia, where many labs held and worked on SARS samples. On December 18, 2003 WHO released a new protocol for handling SARS specimens in the post-outbreak period, with special emphasis on reducing risk of and performing surveillance to detect laboratory infections. Although this protocol was clearly created after the first (Singapore) escape, the WHO chose its words carefully so as to avoid offending members but resolutely highlighted the laboratory risk-factor in its introduction:
“The possibility that a SARS outbreak could occur following a laboratory accident is a risk of considerable importance, given the relatively large number of laboratories currently conducting research using the SARS-CoV or retaining specimens from SARS patients. These laboratories currently represent the greatest threat for renewed SARS-CoV transmission through accidental exposure associated with breaches in laboratory biosafety.”
WHO [t.16]
Post Scriptum (Jan 2022):
On 24th Jan 22, Science magazine published an article about a recent Academic Sinica SARS-CoV-2 leak which revealed the true identity of Lieutenant Colonel (LTC) Chan.
The Science article effectively identified LTC Chan Jiacong [t.21] under a slightly different name: Jan Jia-Tsrong. That revelation, made directly by Jan himself to the article author, actually corroborated a piece published in Global Times not long before [t.21].

A similar incident happened in Taiwan 18 years ago. Taiwan media said that in 2003, the island had a P4 laboratory infection incident. A protagonist named Jan Jia-Tsrong became the island’s last confirmed SARS case, due to the wrong operation regarding the SARS experiment waste.
Global Times, 10 Dec 2021[t.21]
According to his CV, Jan received a honorary fellowship for academic excellence from Johns Hopkins University in 1995 and received his PhD in 1998, in line with an early report in a Chinese source [t.2].
At the time of the Dec 2003 SARS leak, Jan was studying the effectiveness of common and traditional medicines applied to SARS, see for instance ‘Small molecules targeting severe acute respiratory syndrome human coronavirus’ (PNAS, Jul 2004) and ‘Inhibition of Severe Acute Respiratory Syndrome Coronavirus Replication by Niclosamide’ (Jul 04). This is consistent with reports in the Taiwan press at the time.
Jan has recently been doing the exact same kind of research for SARS-CoV-2. See for instance ‘Identification of existing pharmaceuticals and herbal medicines as inhibitors of SARS-CoV-2 infection’ (Jia-Tsrong et al, PNAS Feb 21) and ‘A traditional Chinese medicine formula NRICM101 to target COVID-19 through multiple pathways: A bedside-to-bench study (Keng-Chai et al, Jan 21).
So the twist in the story is that Jan who got infected with SARS in a military P4 in Dec 2003 was supervising the lab worker who got infected with SARS-CoV-2 in a P3 lab in Nov. 2021, possibly while working on the same kind of common/traditional medicine studies.
3. The Ugly: Beijing P3 — February to April 2004
In a few words: The third lab incident is a very unsatisfactory affair. It happened in the context of a rather toxic ivory-tower academic system leading to unchallenged bad practices. Very limited official information was released when it ever was; effectively the Chinese government, and consequently the investigation report, provided very little insights or simply did not discuss key issues.
There are also some reasons to suspect an internal cover up of the first two SARS cases, and last (and not least), the sanctions announced by the Chinese Ministry of Health seem to have been more symbolic than anything else.
Without exaggeration, the Beijing LAI accidents and the subsequent outbreak seem very much to be a story of towering academic ego, shocking incompetence, obstruction of the truth and lack of accountability.
More importantly the unchallenged poor-handling of the investigation by the Chinese authorities — after the WHO left the scene — may have set a bad precedent.
Partially redeeming this dark picture, it is worth noting that the mainland China media contributed some very good pieces of investigative journalism which can still be found today online and deliver many essential clues as to what actually happened.
3.1 Setting the wrong expectations:
Following the Singapore SARS lab accident it became clear that laboratories handling SARS may not be as safe as one could expect, especially in nations rushing to build more P3 labs. This raised the question of the safety of the laboratories handling SARS in China.
Around the 13th Oct 2003 (so before the SARS accident in Taiwan) the National Health and Family Planning Commission of the People’s Republic published an article where it stated that:
“The nation’s SARS virus laboratories are safe. At present, no SARS patient has been infected from a research laboratory virus.”
[b.4, b.13]
But the article went on to list reasons to be anxious and to make sure that the nation redoubles its effort to better control all risk factors:
According to Professor Zhu Qingyu, a P3 qualification only refers to the laboratory’s compliance with physical protection standards. In fact, the human factor comes first in terms of laboratory safety.
“Without good quality and sense of responsibility, (scientific research institutions) without strict and perfect management, being designated a P3 laboratory alone will not work.“
[b.13]
On December 18, 2003, two days after the revelation of the Taiwan SARS lab accident, the WHO called for vigilance in P3 settings studying SARS and issued its ‘biosafety guidelines for handling of SARS-CoV specimens and cultures’ [t.16].
On the same day, the Chinese Ministry of Health duly followed suit and issued an emergency notice requesting all localities to strengthen the centralised management of infectious SARS virus strains and human specimens to ensure the biological safety of virus laboratories and storage units [b.12]. The Ministry of Science and Technology and the Ministry of Health also conducted safety inspections of the 15 to 20 P3 laboratories across the country at the end of December and started addressing the issue of labs that were handling SARS without approval. [b.1, b.12]
Incidentally, the CDC Institute of Virology in Beijing, being the most famous P3 lab handling SARS in the country, was explicitly mentioned in the press at the time:
P3 laboratories have corresponding strict operating procedures. [ — ]. The current research on SARS by the Institute of Virology of the China Center for Disease Control and Prevention is under such a strict operation. SARS laboratories implement a dual access system: first, the procedures must be complete; second, entering the laboratory must be approved by management personnel.
It is reported that in the laboratory of the Institute of Virology of the Chinese Center for Disease Control and Prevention, there are six or seven researchers engaged in SARS research, and these people can be directly exposed to the SARS virus. According to Ruan Li [director of the institute], in accordance to the rules, when researchers enter the laboratory, they must first pass through an air-lock, then through the anti-contamination area, and finally through another air-lock. The same is true when one comes out.
All viruses are wiped out or removed in the process, and they cannot escape the laboratory. Therefore, the key lies in whether the staff strictly follow the procedures. [b.12]

On 19th Dec, Qi Guoming, director of the Science and Education Department of the Ministry of Health of China, detailed the actions taken by his ministry in an interview with a reporter from CCTV:
The Ministry of Health is now dealing with these problems. The laboratories that began to conduct SARS virus research without approval are being cleaned up and checked.
He explained that the review teams will examine the laboratory’s hardware, personnel quality, and the management of virus samples. Those laboratories that do not meet the standards will immediately stop research. [ — ]
‘If an accident occurs and infection is caused, it is a crime against the people and society’ .
[b.12]
3.2 Flagship role of the CDC Institute of Virology:
The National Institute for Viral Disease Control and Prevention (NIVDC) in Beijing (病毒病预防控制所), in short the Institute of Virology, is part of the Chinese Centre for Disease Control and Prevention (China CDC, 中国疾病预防控制中心 ).
The CDC itself had only been formed in early 2002 and had to immediately ride through the SARS epidemic. In June 2003, mainland China had no more new or active SARS cases and the SARS epidemic was formally declared as being over. The Institute of Virology was then designated by the Ministry of Health as one of the six storage units for the SARS virus. [b.1]
The Institute of Virology was effectively the most prestigious viral research institution in the country at the time. It had the first P3 in China, was the workplace of famous academicians and was part of the newly formed CDC, which very purpose was to prevent epidemics in the country.

Situated at №100 Yingxin Street, Xicheng District, the institute is 1 km from the busy interchange station of Beijing metro lines 4 and 7. The main body of the virus institute was a five-story building (since rebuilt), the fifth floor was a dormitory for staff or visitors , and the remaining four floors were offices and laboratories [b.1].
From the appearance, the whole building was unremarkable. An old man who lived in the bungalow opposite the virus institute for more than 30 years told reporters that the virus centre was built in the 1950s [b.1].
To this day the building is still standing. The Institute's main buildings are now at 155 Changbai Rd, Changping Qu, Beijing. But the old building is still part of the Institute. [b.517]
‘№100 Yingxin Street, Beijing still stood stubbornly in that old crowded
alley. The windows of the building were covered with grey dust, many were broken, and the exhaust vents jutting from the laboratory pointed at the residents’ homes only a few meters apart.’
[b.3]


At the time, there was no logo of the Institute of Virology at the front of the office building [b.1]. Its nondescript appearance clearly contrasted with its flagship role in viral research.

3.3 An academic ego got hurt
The director of the Diarrhoea Virus Department was Academician Hong Tao (洪涛) of the Chinese Academy of Engineering. 72 y-old at the time and a highly respected virus expert, he was one of the main pioneers in the field of medical electron microscopy, the main founder of the virus morphology discipline in China, the discoverer of diarrhoea rotavirus (“洪氏腹泻” Hong’s Diarrhoea) which caused a acute diarrhoea outbreak in North China in 1983, the discoverer of the intracellular morphology of epidemic haemorrhagic fever virus, and one of the founders of the Chinese Society of Electron Microscopy which he used extensively in his virus morphology work. He also had extensively studied chlamydia. [b.11]

However, recently his prestige had got rather badly damaged.
At the beginning of the 2003 SARS epidemic that started in Guangdong province, on 13th Feb 2003 precisely, Guangzhou Nanfang Hospital (on the SARS battlefront) issued an autopsy report of a SARS victim, stating that the cause of death was “viral pneumonia.” [b200]
At that very same time Hong Tao and an assistant at the Institute of Virology in Beijing were finding chlamydia particles from lungs tissues of two SARS victims, through electron microscopy.
On 18th Feb the Institute of Virology of the Chinese Centre for Disease Research announced that the main pathogen of atypical pneumonia in Guangdong was likely to be chlamydia. [b.203, b.103]
In the afternoon of the same day, the Guangdong Provincial Department of Health held an emergency meeting. The expert group headed by Zhong Nanshan, a member of the Chinese Academy of Engineering (whose attitude and work during SARS would be exemplary) believed that chlamydia cannot be simply identified as the only pathogen and railed against the limitations of the electron microscope approach.
In effect, Hong Tao had made a basic mistake by ignoring the golden rule of infectious disease pathogen identification: Koch’s postulates. More precisely, he actually ignored 3 of the 4 Koch’s postulates — quite a feat for an academician. [b501]
“If it is chlamydia, I will cut off my head and give it to you… .”
Dr Cai Weiping of the Guangdong expert group , from his hospital bed, after being infected while treating SARS patients [b102]
Both Hong Tao and Zhong Nanshan were members of the elite Chinese Academy of Engineering, but Hong Tao was more senior, based in Beijing, had been in the CCP army back in 49 (very important for connections) and was working for the CDC not for some hospital in the province. Hence, the Chlamydia hypothesis became the official theory in China for a while, and totally ineffective antibiotics treatments were formulated accordingly.
While the world scientific community raced ahead, Chinese researchers who disagreed with Dr. Hong couldn’t get their hands on tissue, blood, and serum samples from SARS patients.
On March 17–18 the CDC and the Ministry of Health held a first national joint meeting of SARS experts. At that meeting, China’s CDC was designated as the country’s leading institution for SARS scientific research, taking the lead back forcefully:
“No unit or individual is allowed to publish research results in any form, including articles; if you want to publish, you must first report to the China CDC.”
[b200]
Incapacitated by that incorrect official theory, Chinese scientists could not publish a single descent paper on the origins of SARS at that time [b301]. In particular it would later emerge that:
- Nanfang Hospital (Guangzhou) had found virus particles in lung tissue samples of SARS patients through electron microscopy as early as February 20, but their research was forced to stop after the “ban” issued at the meeting of the 17–18 March [b200]
- Researchers from the Chinese Academy of Military Medical Sciences (Beijing) also discovered virus particles in samples on February 26, which were identified as a coronavirus after being reviewed by 6 virus morphologists. On March 21, the Military Medical College reported to the relevant departments the discovery of the coronavirus. However, the findings were not officially announced until April 9. [b200, b103]
Eventually, the Chlamydia theory was proven wrong by scientists abroad (mostly US and HK), by the beginning of April it was clear that a coronavirus was the cause of SARS.
Defying that trend, on 1st Apr 2003, Hong Tao and 20 other Chinese scientists published a paper defending the Chlamydia hypothesis [b207]. Nevertheless, on 3rd April 2003 the WHO declared SARS to be likely caused by a coronavirus [b.510], and on 16th April 2003 it confirmed it to be the cause. [b509]
Even after the 16th April, an undeterred Hong Tao would tell reporters:
“It does not mean that this virus is the only culprit. [..] We cannot be sure that chlamydia is an important cause of death for patients, but according to our research on previous cases, the coronavirus alone is not so serious.”
[b.203]
3.4 Fighting back with more P3s doing SARS research
Before the SARS epidemic, in 2002 (as part of the important reforms of the CDC in January of that year), Hong Tao’s Virus Morphology and Viral Diarrhoea Laboratory (病毒形态学与腹泻病毒实验室) at the CDC Institute of Virology was divided into three laboratories, all under his control: the prion (which can cause mad cow disease) laboratory department, the viral diarrhoea laboratory and Hong Tao’s laboratory, designated as the Academician Laboratory (each of the 5 Academicians at the Institute traditionally have their own laboratory). [b.3, b.8]
As discussed above, Hong Tao had recently lost quite a bit of credibility in the “Chlamydia Incident” (“衣原体事件”). After that sorry episode, it was reported that Hong Tao did not go to work at the Institute of Virology often, but concentrated on books at home. Nevertheless, he still had a voice on investment matters and reviewed work (as should be expected of any Chinese academician).
Under Hong Tao the CDC had 3 P3 labs. One was supposed to be working on SARS, but what if these 3 laboratories could be made to work on SARS? Then Hong Tao and the CDC could carve themselves a leading role again in SARS research.
Let’s remember that at the beginning of 2004 the number of P3 labs in the country was still very limited, at around 15 to 20. Because of security concerns [b.4], the few SARS virus specimens that were necessary for experiments had become a “scarce resource” and were available only at the 6 designated storage institutions — which included the Institute of Virology [b.1]. So in effect very few P3 labs in the country could do SARS research on unattenuated live strains, and with a bit of administrative flexibility three of them could be right under Hung Tao at the Institute.
Wang Jianwei (王健伟) who until then as an associate researcher at the Institute became the director of the viral diarrhoea department around Sep 2008. He was a former doctoral student of Academician Hong Tao and as an assistant, he was the one who with Hong Tao first discovered chlamydia in the lung tissues of deceased SARS patients. [b.3]
From his narrow field of viral diarrhoea Wang Jianwei could soon move to research on the much more critical SARS virus, with the encouragement of Hong Tao:
“Wang Jianwei is my student and I have the responsibility of teaching. SARS still has too many mysteries.”
Hong Tao, explaining his work on SARS after the Chlamydia Incident [b.3]
As one Chinese article would soon put it:
This relation between Hong Tao and Wang Jianwei explains exactly why post-doctoral students in the diarrhoea virus lab could do “interdisciplinary research on SARS virus” and select SARS as a major. For young researchers, the mysterious SARS virus is not only a severe challenge, but also a rare opportunity.
[b.3]
The source above [b.3] goes on by reporting an official description of these students as having no weekends and holidays, no day and night, and sometimes even working overnight in the BSL-3. Good traditional party-speak, but not actually the best advertisement for lab-safety if one thinks of it.
As a result of this ‘interdisciplinary research’ approach, while the virus emergency technology department and the virus resource centre department were technically responsible for the preservation of SARS strains, some SARS strains and potentially infectious materials were also kept in the diarrhoea virus and in the prion virus laboratories.
3.5 Lab contamination — Feb 2004
Ren Xiaoli (任 小 莉, pseudonym) is a Virus Morphology doctoral student at the Institute of Virology in his last year before graduation. Under the guidance of Hong Tao (founder of the Chinese Virus Morphology discipline) and Wang Jianwei (王健伟), then director of the Viral Diarrhoea Department of the Institute of Virology, Ren Xiaoli and 21 doctoral and master students such as Yang (杨某) and Guo (郭某) joined the frontline of SARS scientific research. [b.3]
To understand exactly what happened we need to understand the layout of the rooms accessible to the viral diarrhoea department members. Unfortunately, the official reports are very opaque and somewhat confusing as to these essential aspects. Fortunately, some key insights were provided by insiders to reporters of Caijing and Southern Weekly [b.15]. Without these, it would be impossible to make any sense of the limited official reports.
The viral diarrhoea department rooms seem to have been divided between P3 rooms and ‘normal’ rooms (possibly BSL2 [b505]). In particular one of the ordinary rooms held an electron microscope, a large apparatus that was the favourite investigation tool of Hong Tao and of his doctoral students.
Rooms 106 and 107 (on the first floor of the main building of the Institute of Virology) were mentioned by insiders. Presumably one room was the P3 laboratory, while the other one next door was a P2 electron microscope room.[b.1]
A professor in the Department of Medicine at Peking University, who has repeatedly entered the China Centre for Disease Control, told reporters that the management of the virus has not been strict enough. For example, the strain was originally required to be stored in a special safe in the laboratory, and locked, but at some stage the SARS strain was placed outside in the corridor because the overcrowding of laboratory personnel was very serious. According to his estimate, the number of laboratory staff at that time was three times the reasonable load of the laboratory. [b.4]
‘because the room was too crowded, the refrigerator containing the SARS virus was moved into into the aisle’
[b.3]
In other words because the (likely newly established) P3 lab room of the viral diarrhoea department was too crowded, the samples fridge of the diarrhoea room was moved into the corridor, by the entrances to the 106 and 107 rooms. Consistent with improvised security measures to keep that fridge safe despite having it standing in a corridor, we are further told that
‘there is a padlock on the refrigerator door, and the key is kept by two people. To be on the safe side, a seal was also affixed to the refrigerator.’
[b.1]
This interpretation is in line with one of the first notices issued by WHO when responding to the leak accident:
’investigators have serious concerns about biosafety procedures at the Institute — including how and where procedures using SARS coronavirus were carried out, and how and where SARS coronavirus samples were stored.’
WHO, May 18 [b503]
After the Spring Festival holidays in 2004, Ren Xiaoli regularly took out the kit containing the normally attenuated SARS virus from the phosphate PH buffer solution in the fridge belonging to the P3 viral diarrhoea lab (but standing in the corridor) and directly walked into the ordinary Electron Microscope room next door [b.3], as she needed to use the electron microscope equipment there — a typical pathogen investigative approach of Hong Tao’s students.
Some report states that Ren actually prepared the deactivation solution [b508]— but being a student the method itself would likely have been decided by a superior.
She never thought that the dangerous virus in her hand might still survive. Indeed, According to previous experience, a solution formulation composed of detergents such as phosphate buffer and sodium lauryl sulfate can deactivate the active protein of the virus. However, the SARS virus inactivation technology adopted by the viral diarrhoea department had not been effectively verified, and it was not included in the SARS virus inactivation program recommended by the Ministry of Health.[b.3]
Unfortunately, this would cause a contamination of the ordinary electron microscope room. Ren and Cui (a virus morphology student in the viral diarrhoea department) developed symptoms such as diarrhoea and high fever as early as the 8th February 2004 [b.1, b.8], and went to Union Hospital and Friendship Hospital successively. Fortunately, their condition improved later and they did not infect anybody. [b.3]

At this point, we must note that Ren and Cui cases only publicly emerged when they were found to be positive for IgG and IgM SARS antibodies in May 2004 [b.1], during the investigation triggered by the April LAI when all employees were tested.
China only succinctly mentioned their cases and the role they played in early July 2004 [b58], so about 2 months after they were first disclosed by Caijing. Shortly after that short official mention, an investigation by Southern Weekly [b.3] added more details obtained from insiders.
That’s all the information that was ever available on these two cases — despite them being essential for the rest of the story, and the names are pseudonyms. Additionally, these two crucial cases will not be included in the final Chinese report [b51]. Nor would the WHO ever mention them at the time [b501], thus mirroring the Chinese report.
In the end it is not clear whether the Institute of Virology itself actually knew about these cases before May when the Ministry of Health organized some systematic antibodies testing, but the question may be worth asking especially since these two researchers checked into some hospitals.
3.6 Community Transmission — Apr 2004
On March 7th Song (宋某 as given in the reports at the time), a 26-year-old graduate student of Anhui Medical University (安徽医科) started a short-term internship in the viral diarrhoea department of the Institute of Virology in Beijing, during which time she worked with adenovirus (腺病毒) and syncytial virus (合胞病毒). Her research has nothing to do with SARS. With her qualifications, she was not allowed to enter the P3 laboratory for SARS research, and she always maintained that she never did. [b.1, b.4]
On the evening of March 23, she took the train back to Hefei (合肥, her hometown, a 1,000 km train journey). On March 25, she felt body aches, fever, and physical discomfort, she took cold medicine and antibiotics by herself, but her symptoms did not alleviate. She returned to Beijing by train on March 27, and went for a consultation at Beijing Jian Gong Hospital (健宫医院) on March 29 where she was admitted for pneumonia. [b.1]
Inadvertently, she has been infected with the SARS virus.
Her mother Wei (魏某) came over to Beijing from Anhui on Mar 31 to be with her and, if possible, bring her back home. On April 2, Song and her mother returned to Hefei Huainan (Anhui) by train, where Song was transferred to the Second Hospital of Huainan Mine (淮南矿二院). According to Caijing, the Virus Institute did not doubt Song’s symptoms at the time, but “later her mother took her to Anhui” and the Institute did not continue to investigate [b.1].
“She was actually very poor [ — ]. Sometimes I even recalled the way she was lying on the hospital bed, her fever was terrible, the expression on her face and the pain in her eyes. But I just feel that she was too stubborn and did not cooperate with our treatment, saying that she was going back to Anhui to continue treatment.”
Nurse Li, to a reporter [b.6]
On April 4, Song was further transferred to the First Affiliated Hospital of Anhui Medical University (安徽医科大学附属第一医院) for viral pneumonia to continue treatment.
On April 8, Song’s mother, Wei, who had now been with her for 8 days, began to develop fever and was admitted to the First Affiliated Hospital of Anhui Medical University with unexplained viral pneumonia. On April 19, Wei’s condition suddenly worsened and despite efforts to save her she died (community case #1). After receiving the report, the local health department immediately activated the SARS early warning mechanism. [b502]
Since she had never been exposed to this deadly virus, Song did not receive any warning. Even after the death of her mother, Song did not think of the word “SARS”. From beginning to end, Song never suspected that he was infected with the SARS virus.
“I have never been to a laboratory that stores the SARS virus.”
“I still don’t know when and where I was infected with the terrible SARS virus”
As told by Song [b.2, b.14]
At Beijing Jiang Gong Hospital, a 20y old nurse named Li (李某), whose job was to take care of fever patients not yet examined, had been in contact with Song [b.6]. On April 5, nurse Li began to experience chills while waiting for a bus. On Apr 7, she developed fever, cough and other symptoms, and was admitted to Jian Gong Hospital .
Because her condition did not improve after treatment, she was transferred to the intensive care unit of Peking University People’s Hospital (北京大学人⺠医院) on the 14th. She first learnt that she had contracted SARS on 21st April. [b.1]
Song was not the only primary case. Yang, a post-doctoral fellow at the institute [b502], fell ill after working in the viral diarrhoea department. He was hospitalized 23 days after Song (17th April) and then put in isolation on 22nd April on confirmation of the SARS diagnostic.
3.7 The Response of the Authorities
China officially reported Li’s SARS case on 22nd April, then Song, Wei’s and Yang’s cases on 23rd April [b502, b504]. The institute was temporarily closed and quarantined on 23rd April. [b1, b513]

An additional case was reported on 28th April, a 49y old female retired doctor who on 12th April was in the same hospital ward as nurse Li. In total, Li was linked to 5 confirmed cases. [b.7]
During the 2003 outbreak, the transmission of SARS was greatly amplified in hospital settings. As a risk reduction strategy, all seven Beijing SARS cases were eventually treated in one selected hospital, Ditan Hospital (with Song being eventually treated in Anhui).
Nevertheless, the patients were initially treated or assessed in open wards at seven hospitals (five in Beijing and 2 in Anhui) before suspicions of SARS were raised and procedures of isolation and infection control were introduced. In addition, the two patients in Anhui travelled long distances within China by train. As these events created opportunities for multiple exposures, Chinese authorities undertook extensive tracing and follow-up of contacts.
By the end of April, 700 people were medically quarantined. More than 260 people from the Institute of Virology, including scientific researchers, graduate students and their family members, were quarantined or dispersed, with 24 people being sent to the Beijing Chest Hospital. In the town of Xiaotangshan, Changping District, a resort received 145 people. [b1]
In total, nearly 1,000 people would be quarantined (a figure never released by the Chinese CDC). [b106]

Two additional confirmed cases of SARS and three additional suspected cases were reported in Beijing on 1 May, all related to the Viral Diarrhoea Department of the CDC’s National Institute of Virology in Beijing.
“The cases had been linked to experiments using live and inactive SARS coronavirus in the CDC’s virology and diarrhoea institutes where interdisciplinary research on the SARS virus was conducted.”
China Daily, [b.59]
On 2 May, China announced the three suspected cases as genuine cases of SARS, bringing the total cases in a recent outbreak to nine. 189 people were released from quarantine.
On 18 May, after no new infections had been reported in a three-week period, WHO declared that ‘China’s latest SARS outbreak has been contained, but biosafety concerns remain”. [b503]
On May 21, Zhang, the last SARS case in Beijing, was discharged from Beijing Ditan Hospital.
On May 23, all 747 close contacts in Beijing were lifted from quarantine, the treatment of SARS patients in Beijing and Anhui ended and the SARS transmission chain was considered as stopped. [b.54]
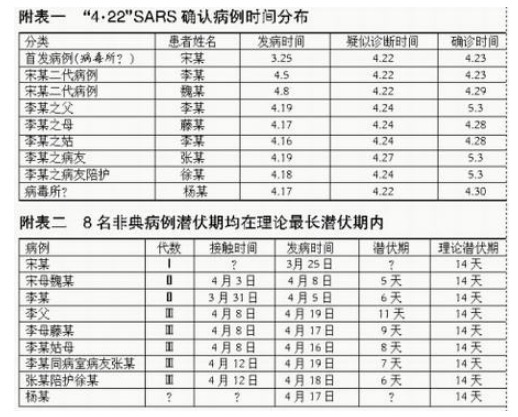
3.8 Summary of cases:
There were in total 11 cases over four generations. Note that the official report would only mention the 9 cases of the Song-contamination chain (April), ignoring the two February primary cases that are linked to the contamination of the diarrhoea lab and had been disclosed by Caijing since May. [b512, b513, b514]
a. February Lab contamination: 2 primary cases:
Ren (pseudonym) and Cui were found positive for antibodies in May. According to the official report, Ren caused the contamination of the Viral diarrhoea department by taking the unattenuated SARS virus from the sample fridge lab for observations under electron microscope. Little is known about Cui beyond the fact that he worked in the viral diarrhoea department. It was established that he contracted SARS not from Ren but independently, making him another primary case.
For some reason, neither Ren nor Cui are included in the list of patients in the official investigation and WHO reports at the time — which instead focuses on the April outbreak. They will eventually be mentioned by the WHO in [i.1].
Given that these two cases ended up in hospital, it is rather likely that they raised an alert and that the LAIs were detected at the time, and not after the blanket IgG testing of staff and students following the April outbreak. The cover-up by the Chinese authorities of these two early cases, which persisted even after the April outbreak, can then be explained by the rather unsavoury reasons for the contamination of the institution.
When eventually forced to publicly deal with the April outbreak, the Chinese were then careful not to ever mention these key February cases, leaving the reason for the April outbreak officially unresolved when that reason was by then well known (and even leaked to Caijing).
b. April events: 1st generation: 2 primary cases from contaminated lab
Song (宋某), a 26-year-old female postgraduate student from Anhui Province. She had started an internship in the viral diarrhoea department on 7th Mar. She did not return after the 22nd March as she soon fell ill.
Yang, a 31-year-old male post-doc researcher at the same virology institute who was hospitalized on 17 April, got independently infected. Indeed, he started developing symptoms about 15 days after he last met Song, briefly in a corridor of the Institute, when the incubation period of SARS is 2 to 10 days. [b7, b516]
c. April events: 2nd generation.
The two cases in the second generation are both linked to close personal contact with the postgraduate student. These cases are her 53-year-old mother, Wei (魏某). The second case is a 20-year-old nurse in Beijing, Li (李某), who treated the postgraduate student, from 29 March to 2 April, during her initial hospitalization in Beijing.[b.7]
d. April events: 3rd generation.
Five further cases have all been linked to close contact with the nurse. Three are relatives: her 45-year-old father, her 44-year-old mother, and a 36-year-old aunt who visited her in hospital [b515, b.7]. The fourth case linked to the nurse is a 49-year-old retired female doctor who was admitted to hospital because of another illness and shared a room with the nurse. The retired doctor’s 23-year-old daughter-in-law, who accompanied her at the time of hospital admission, is the fifth case. [b.6]
3.9 The Official Investigation
a. Brief role of the WHO and missing February cases
The WHO sent a team on 26th April 2004 which included ‘experts in epidemiology, virology, infection control, and laboratory biosafety’ [b518].
It seems that the WHO team was supposed to second the Chinese investigative team, not to do its own independent investigation:
‘On April 23, the Ministry of Health established an expert group composed of the Academy of Military Medical Sciences, Beijing Centers for Disease Control and Prevention, and China Centers for Disease Control and Prevention on infectious diseases to conduct a special investigation on the source of infection in this SARS case. WHO sent experts to participate in the investigation.’ [b519]
On 12th May, that expert group formed an investigation team composed of 7 experts from the Academy of Military Medical Sciences, Beijing Municipal Centre for Disease Control and Prevention, and CDC Institute for Infectious Disease Control and Prevention was formed. [b.1, b.3]
This China-WHO team conducted a thorough review of the April cases (but not of the February ones who were officially discovered sometime in May via systematic IgG testing of lab workers [b1]):
‘The investigation team conducted epidemiological investigations on the two [April] cases, interviewed all the staff in the same laboratory, conducted on-site investigation and sampling of the virus, and handed the collected samples to two national-level laboratories and one. The WHO network laboratory conducted tests according to a unified technical plan. As of May 11, the expert team has not yet determined the source of the infection.’ [b519]
Note that ‘the expert team has not yet determined the source of the infection’ is disingenuous. First it had been established by that time that it was a case of lab contamination [b518], so really what is meant is that the precise source of the lab contamination is still unknown. Secondly, understanding the source of the contamination requires looking into the two February cases identified by testing in May — which the Investigation team was not doing.
‘Results of investigations to date point to laboratory research at the National Institute of Virology in Beijing as the likely source of the outbreak. The institute has been engaged in research with the SARS coronavirus, including the development of a vaccine’. [b518]
On May 14, it was learned from the representative office of the World Health Organization in China that the investigation team had confirmed that the SARS epidemic originated from a laboratory infection at the Viral Disease Control and Prevention Institute of the Chinese Centre for Disease Control and Prevention (CDC), but the investigation team still did not clarify the details.
On May 21, Caijing published the first article in the Chinese press [b1] mentioning the lab escape and the early February cases. Hence, the Chinese side was actually aware of these early cases at the very latest by the 21st, probably some days before.
The last WHO update was on 18th May 2004. No update followed and no WHO report was released. The investigation was effectively to be managed by China.
b. The Official Conclusions
The Ministry of Health would produce its investigation report on 1st July 2004. There is no mention of the WHO at the time in the press reports. The WHO team had most likely long left the scene and its role in the investigation, whatever its extent, was not discussed.
The Ministry of Health would produce its investigation report on 1st July 2004 [b51], under the title ‘Analysis Report on the Epidemic Control of the ‘4.22’ SARS Epidemic in Beijing in 2004' (2004年北京‘4·22’SARS疫情流行控制情况分析报告).
The report covers the nine April cases starting with Song. It briefly mentions Ren & Cui by stating that they had tested positive for IgG and IgM antibodies but does not include them with the nine April cases and does not mention that their cases go back to early February. [b58]
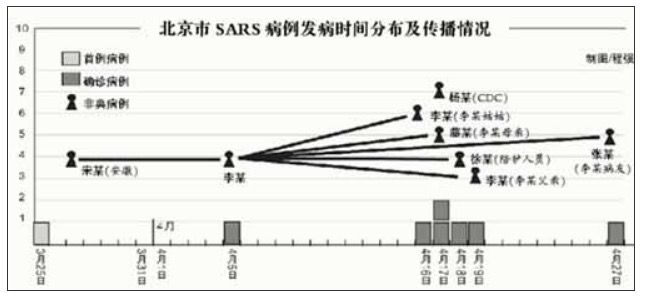
The investigation pointed out the six major problems of “illegal working procedures”: [b100, b58]
First, research projects are interdisciplinary . The viral diarrhoea department researches the gastrointestinal virus field, but has undertaken SARS research projects as a cross-disciplinary topic. The staff are not familiar with the topic, which has caused safety risks.
Second, safety management is not paid attention to. The staff adopted a new method of inactivation, without the academic committee’s validation, which scientific basis was insufficient, and the department director approved it without authorization. The inactivation effect had not been strictly verified, and there is no verification plan, record and content.
Third, technical operations are not conform to the standard, as per the absence of safety precautions in ordinary laboratory operation, thus violating the regulations of the Ministry of Health that the inactivation of SARS virus must be carried out in a laboratory with a biosafety level above P2 or in a biological safety cabinet.
Fourth, the staffing is not strict. A large number of graduate students and fellows who lack professional knowledge are used for high-risk research, and there is no strict biosafety knowledge training for relevant personnel.
Fifth, health monitoring is not in place, it violates the laboratory personnel health monitoring system, accident reporting system and other regulations formulated by the Ministry of Health, failed to promptly report abnormal conditions such as multiple fevers of laboratory personnel, and failed to take necessary measures. Two of the laboratory staff developed fever, and one was hospitalized for two weeks, without attention being paid to the cases and no reporting was made.
Sixth, the implementation of the system was not serious and violated the rules of the Ministry of Health and other four ministries regarding the implementation of two-person access to the P3 laboratory, as single-person operations occurred many times.

Investigative journalists add important elements
Over about two months, a few Chinese magazines and newspapers were able to add some crucial elements of this story (some acquired via insiders), such as the positive cases of Cui and Ren [b1] and the location of the fridge out of the P3 lab [b3[.
3.10 The omissions
Some international experts were disappointed that many details about the incident and the lab’s operating procedures remain very opaque or simply hidden.
“I was hoping for a full, more open account of what happened,”
Antony Della-Porta, the Australian biosafety expert who led the earlier WHO investigations of the SARS escapes in Singapore and Taiwan, but not Beijing. [b508]
a. No full report published
This was supposed to be a joint China-WHO investigation [b519].
However, the WHO simply withdrew from the scene without trace. There was not even an update posted by the WHO after the 18th May, and the WHO did not publish the official report produced by China.
China itself only made public a few brief extracts on 1st July 2004 (extracts which do not mention the WHO). It never published the full report.
The limited role of the WHO was confirmed in a WHO press release date 2nd July 2004 [b59]:

b. February inspection of the Institute
On July 12th, 2004, shortly after the reorganisation meeting, according to media reports at the time, the Ministry of Health was rather sanguine about the SARS virus leak. And they had good reasons.
The investigation report obtained by some reporters indicated that in February of that year (2004), following the new SARS-samples handling protocol issue by the WHO on 18th Dec 2003, the leaders of the Ministry of Health went to the Chinese Centre for Disease Control and Prevention to inspect the P3 laboratory of the Institute of Virology and found problems with it. They asked the CDC to propose a rectification plan and a plan to provide financial support, but the CDC did not implement it seriously. [b106]
Possibly, the inspection was just part of the Ministry review of P3 labs triggered by the new WHO guidelines following the Taiwan lab escape — however most P3 labs are understood to have been visited at the very end of December 2003 or early in January. Alternatively, it may have been triggered by the two suspected cases of SARS (Ren and Cui) who ended up in hospital in February.
Whatever the reason, the Ministry of Health and the Chinese Centre for Disease Control and Prevention knew of some issues at the laboratory back in February.
c. The cause of the contamination
Ren and Cui had tested positive for antigens back in May, as disclosed by Caijing [b1]. Judging from the few available extracts, the Ministry of Health report simply confirms the positive tests but never mentions the February context, nor does it include them in the official count of the cases (hence stopping at 9 and not 11).
In any case, the official name clearly focuses on the April chain of contamination (“‘4.22’ SARS Epidemic in Beijing in 2004”) [b51] so implicitly dropping the early February cases.
As mentioned above, the purpose of this sleight-of-hand was most likely to avoid having to disclose the rather unsavoury reasons of the contaminations at the institute.
3.11 The sanctions:
The Ministry of Health decided the following sanctions: [b4]
Ruan Li (阮力), director of the Virus Disease Control Institute who has the main leadership responsibility, Dong Xiaoping (董小平), deputy director of the Institute, and Wang Jianwei (王健伟), director of the Viral Diarrhoea Department, received administrative sanctions. [b4]
Li Liming (李立明), the director of the Chinese Centre for Disease Control and Prevention was given administrative demerits and resigned.[b4]
“Neither Taiwan nor Singapore’s laboratory infections caused second-generation cases, but this time our SARS incident not only caused transmission, but also third-generation cases. This is our failure to do a good job. Sorry to the government and people. We don’t have any excuse on the issue.”
Li Liming, May 8 2004, at a reorganization meeting held by the Institute of Virology
Yang Xiaoguang (杨晓光), his deputy director, who had important management responsibilities, was given major demerits and resigned. [b.4]
As for the student behind the pseudonym of Ren Xiaoli, we are told that s/he was not punished, being only a student and not directly responsible. This seems a bit odd and some questions are worth asking about the true identity of that person — given as the main proximal cause of the contamination of the lab — if the story concerning that person is true indeed.
Academician Hong Tao, director of the Institute, was not sanctioned. This triggered some rather vitriolic analysis of the nepotism of the Chinese scientific institutions:
“The academician system has also set an example for new academics in China: Those who work hard and do research are idiots, and those who can get nothing, or get more with less, are ‘brilliant people with leadership skills’.”
Yi Ming, The problems of Chinese academia and their way out (亦明, 中国学术界的问题及其出路), Jul 2004 [b301]
3.12 All is well that ends well:
In any case, the sanctions seem to have been more on paper than anything else:
Wang Jianwei (王健伟) CV states that he worked at the Institute until Feb 2006, then moved to be director of Merieux Laboratory in Beijing, He is today the director of the Chinese Academy of Medical Sciences & Beijing Union Medical College. He also served as a member of the Standing Committee of the Party. Last, he was appointed executive editor of Biosafety & Health magazine [b303] and has produced a manual on laboratory biosafety. [b304]
Dong Xiaoping (董小平), saw his job description change from ‘Director of the Prion Division, Deputy Director, Institute of Viral Disease Control and Prevention, Chinese Center for Disease Control and Prevention’ to ‘Director of the Prion Department, Institute of Viral Disease Control and Prevention, Chinese Center for Disease Control and Prevention’ — so basically dropping the ‘deputy director’ title. In 2012 he would reclaim that full title and is still in the same position. He enjoys special allowances from the State Council and is a member of the CPPCC National Committee (China top political advisory body).
He is also the deputy director of the State Key Laboratory of Infectious Disease Prevention and Control and the director of the Prion Department of the Viral Diseases Institute. He is also a member and chief reviewer of the Biosafety Professional Committee of the National Accreditation Committee of Chinese Laboratories and, finally, he is co-Director of the Chinese CDC.
In February 2020, he became an expert of the “WHO-China Coronavirus Disease Joint Expert Investigation Group” and was part of the February 2020 joint-mission. [b305]

Li Liming (李立明) would later serve as Party Secretary and Executive Vice President of Beijing Medical Association. He is a professor at the Peking University School of Public Health.
Ruan Li is still a professor of the Institute of Virology.
Hong Tao is still a well-respected academician and enjoys special allowances from the State Council.
In 2016, Hong Tao, Wang Jianwei and Song Jingdong published the ‘Medical Virus Atlas’, compiled from nearly 50,000 electron microscope images of viruses.
In 2019 Hong Tao received a commemorative medal for the Celebration of the 70th Anniversary of the Founding of the People’s Republic of China, mentioning his significant contribution to the cause of medical virology and infectious disease prevention and control in China. [b306]
On 7th Jan 2020, his student Song Jingdong (宋敬东), associate researcher at the Institute, took the first-ever Electronic Microscope picture of SARS-CoV-2.
3.13 Déjà vu:
On 5th August 2021 US Right To Know released some emails showing that some senior researcher got infected with SARS-CoV-2 at the very same Beijing Institute of Virology (NIVDC) in January or February 2020:
The SARS-CoV-2 lab-acquired infection came to light in a set of emails dated Feb 14, 2020, between virologists Shan-Lu Liu (Ohio State University), Lishan Su (then of the University of North Carolina) and Shan Lu (University of Massachusetts Medical School).
Shan-Lu Liu noted that his former director at NIVDC “has now been infected with SARS-CoV-2”, and in a separate email acknowledged that his former colleague “was infected in the lab!”
There is a particular irony in the fact that this information was disclosed while Shan-Lu and Lishan were working on a commentary which very purpose was to refute the possibility that SARS-CoV-2 may be a research related accident!
The context of the email exchange was in the preparation of a commentary to refute the hypothesis that the novel coronavirus SARS-CoV-2 came from a lab, which Shan Lu had solicited as editor-in-chief of Emerging Microbes & Infections (EMI), a China-linked journal.
While there was no official reporting of that LAI, at least Shan-Lu recognised the fundamental issue, which is effectively a repeat of the 2004 SARS-1 leaks:
Shan Lu responded, “I actually am very concerned for the possibility of SARS-2 infection by lab people. It is much more contagious than SARS-1. Now every lab is interested in get[ting] a vial of virus to do drug discovery. This can potentially [be] a big issue.”
4. Conclusion: Lessons from SARS Lab-Escapes
4.1 Importance of people and processes:
All the three SARS lab-leaks that we looked at were the result of people either making mistakes or taking deliberate short-cuts, and were often made worse by bad processes that only surfaced after the investigation and were not questioned before — even when seemingly quite obvious to an external observer.
It is therefore important to remember that a P3 or P4 certification is largely secondary in terms of biosafety. The major risk factors are the people entering the lab and the processes that are applied there. It is thus straightforward to build a P3 and then to practically run it as a P2, as far as people and processes are involved — a point that the 2004 Beijing SARS lab leaks amply illustrated.
Compared to other industries, such as nuclear industries for instance, the situation is worse. People working in nuclear power stations typically go through lengthy qualifications. Also, a radioactive exposure or increased coolant temperature can usually be immediately detected and measured via sensors. In contrast, biological P3 labs often see students with limited qualification in biosafety working there (see Singapore and Beijing leaks), and pathogen exposure is typically not detected until too late. The parallel would thus be a nuclear power station where students are routinely given a chance to manipulate radioactive fuel, or put in charge of reactor controls with limited oversight and limited sensors — all after a short risk induction.
In the extreme, a P3 or P4 designation may actually be counter-productive if it leads to a false sense of safety. This was actually very well understood by some Chinese experts straight after the Singapore Lab-leak:
Hidden non-hardware dangers of P3 laboratory:
While the World Health Organization puts the emphasis on the P3 biosafety level, many experts interviewed believe that P3 classification is only one aspect of laboratory safety control. Compared with the laboratory’s hardware facilities, processes (software) and management issues are more important.
According to Professor Zhu Qingyu, a P3 laboratory only means that the laboratory meets the standards in terms of physical protection. In fact, in terms of laboratory safety, the human factor comes first. “(Scientific research personnel) do not have good quality and sense of responsibility, (scientific research institutions) do not have strict and perfect management, and P3 laboratory alone will not work.” Zhu Qingyu said.
WHO warns of hidden dangers in laboratories. Are the nation’s SARS laboratories safe? Oct 2003, Oriental Morning Post — a prophetic article before the Beijing SARS leaks.“Safety is no small matter. A safety breach may occur anywhere.” Gao Fu, an academician of the Chinese Academy of Sciences and deputy director of the Chinese Center for Disease Control and Prevention, said of the accident, “the string of biosafety cannot be relaxed for a moment.”
China Science News (2014–07–25 14th Edition)
A consistent and systematic training is effectively the cornerstone of a lab biosafety,
“These narratives of escaped pathogens have common themes. There are unrecognized technical flaws in standard biocontainment [ — ].The first infection, or index case, happens in a person not working directly with the pathogen that infects him or her, as in the smallpox and SARS escapes. Poor training of personnel and slack oversight of laboratory procedures negate policy efforts by national and international bodies to achieve biosecurity [ — ].”
Martin Furmanski in the Bulletin of the Atomic Scientists, Mar 2014
As to whether this state of affair can ever be fully rectified given human nature, some experts such as Martin Furmanski, remain sceptical:
Looking at the problem pragmatically, the question is not if such escapes will result in a major civilian outbreak, but rather what the pathogen will be and how such an escape may be contained, if indeed it can be contained at all.
Martin Furmanski in the Bulletin of the Atomic Scientists, Mar 2014
4.2 Importance of Laboratory location:
The location of major labs (P4s and top P3s), especially civilian ones, tend to be often in major cities — close to universities and related biological industries — and thus often in the middle of land and air transport hubs, including international transports.
While the advantages of such locations in terms of research and industries are obvious, these locations may also dramatically enhance the risk of a lab breakout being able to move from an undetected limited spread — where the pathogen may die out without having the time or the occasion (location/optimal host) to adapt — to a full-blown epidemic and even pandemic.
The Taiwan and Beijing lab leaks offered some prime examples. In the case of Taiwan, the infected individual flew to Singapore the day after getting infected (so not infectious yet) and then flew back to Taiwan when potentially infectious. Luckily nobody on his flight or in the conference he attended in Singapore got infected. In the case of Beijing, one infected individual travelled back and forth by train three times between Beijing and Ahnui, triggering cases in these locations.
This location risk has been researched and simulated, providing interesting insights. For example, Merler et al modelled the possible spread of a lab escape in various European urban settings (Rotterdam, London, Stockholm, Milan, Madrid and Paris) using an agent simulation and a range of R0. Their conclusion was that the location of the laboratory may play an essential factor in determining the risk of an outbreak based on a lab escape event:
For instance, given a BSL facility located in the UK, we found that the probability of epidemic outbreak when the pathogen is accidentally released from a hypothetical BSL laboratory in Wales (UK) may be three to five times lower than that estimated for a BSL laboratory in London if R0 <1.5. These differences are ascribable to differences in population density and sociodemographic structure [ — ].
“Containing the accidental laboratory escape of potential pandemic influenza viruses” (Nov 2013), Merler et al.
One of their conclusions was that escapes may lead to a cryptic local spread that simply dies out before even being detected. Hence, it is quite possible that in a sparsely populated environment a lab escape may simply trigger an undetected local spread (not much different from a local zoonosis in a remote village), while having a good chance of triggering a full breakout in a much densely populated urban setting in close proximity to transport hubs.
Martin Furmanski additionally drew attention to the simultaneous development of increased pathogenicity studies, which have the potential of releasing a virus with a high R0 — another key risk factor:
Experiments that augment virulence and transmissibility of dangerous pathogens have been funded and performed, notably with the H5N1 avian influenza virus. The advisability of performing such experiments at all — particularly in laboratories placed at universities in heavily populated urban areas, where potentially exposed laboratory personnel are in daily contact with a multitude of susceptible and unaware citizens — is clearly in question.
If such manipulations should be allowed at all, it would seem prudent to conduct them in isolated laboratories where personnel are sequestered from the general public and must undergo a period of exit quarantine before re-entering civilian life. The historical record tells us it is not a matter of if but when ignoring such measures will cost health and even lives. Perhaps many lives.
Martin Furmanski in the Bulletin of the Atomic Scientists, Mar 2014
____________________________________
Box: Can lab processes ever be fool-proof?
Formal processes school:
One standard reaction when faced with human errors and faulty processes is to try to redesign processes and reinforce training. While this answer has some value, experience would indicate that in a domain where the human factor is very important — such as in P3 lab — such an approach may never work perfectly.
For instance:
- Adding more rules for those handling pathogens won’t help if the people infected are usually not the ones handling the pathogens (i.e: indirect infections via contamination of the lab surfaces, via aerosols or via wastes).
- Adding more federal and international regulations won’t help if the regulations aren’t consistently followed.
- If there are still unrecognised technical flaws in the standards for biocontainment, how would we know until an incident made those flaws apparent?
The main issue is that this response draws from a largely mechanical approach to risk which is better suited to industrial environments where (i) the hardware is the main risk factor and (ii) where the human actions are more predictable and standard; and (iii) the human factors do not routinely involve students and other lightly qualified personnel — but highly trained professional elements.
That mechanical approach to risk is also better adapted to an environment where the risk is better visualized: be it mechanical (machineries) or detectable (nuclear radiations, chemical vapours).
Another issue correctly pointed out by Lipsitch and Inglesby as well as others, is that the mechanical approach to risk may also produce incorrectly small evaluation of the risk involved, by ignoring ‘model risk’, the risk that the (often mechanical) model used to derive such risk evaluation is actually incorrect.
[ — ] when one performs a risk analysis and estimates an exceptionally low probability (P) of a catastrophic outcome, it is crucial to consider the probability q (which may exceed P) that the model used to derive that probability is itself wrong, in a way that understates the true probability of the outcome.
[Reply to “Studies on Influenza Virus Transmission between Ferrets: the Public Health Risks Revisited”, Lipsitch and Inglesby]
That largely mechanical approach was used in particular for the Gain of Function Risk Benefit Analysis Report by Gryphon Scientific, which was instrumental in lifting the GoF moratorium in 2017 (a simulation over event trees, with some inspiration from the nuclear power generation risk analysis and the military and civil aviation accident analysis).
Inherent safety school:
An alternative approach to risk is more pragmatic. Drawing on the recognition of the limitations of the human factor, that approach focuses instead on limiting the inherent risk factors.
- Limiting the work on enhanced pathogens (reducing the R0)
- Positioning labs in sparsely populated environments and far away from transport hubs
- Possibly implement practical quarantines in labs
See for instance ASM Letter ‘Assessing and Managing the Risks of Potential Pandemic Pathogen Research’ (Daniel Rozell).
An analogy:
Interestingly, a similar problem was long at play in the world of finance, with a derivatives industry swearing by a mechanical approach to risk which was plagued with modelling failures, and fighting hard to remain lightly regulated. Until the events of 2007–08 that is; events which resulted in better regulation and a return to inherent safety principles (exchange traded derivatives vs. over-the-counter ones, higher provisioning requirement pushing for less inherent risk in the books of the banks, etc).
That it took the near destruction of Western economies to remediate the problem is rather humbling.
_____________________________________
4.3 Importance of Institutional Factors:
The quality of the response to a Lab-Escape in a given country is largely framed by a few key institutional factors, as nicely demonstrated by the 2003–04 SARS lab-leaks:
- Government (central and local) transparency
- Central government willingness to learn from mistakes
- Free press that can contribute to transparency
In particular, we note that:
- The answer in Singapore was very strong on (1) and (2), much less on (3) where the press did not add any material knowledge.
- The answer in Taiwan was very strong on (2), a bit less on (1) most likely due to the military setting (a P3 lab with a military P4 lab), and rather strong on (3) [interestingly mainland China media reports also added some important details].
- The answer in Beijing was very poor on (1) and (2) but rather good on (3), as investigative journalism provided essential details without which we would have only the most limited understanding of the exact circumstances of the leaks, since neither the WHO nor Beijing released any report.
Unfortunately, SARS-CoV-2 outbreak would in due time provide an example where the government answer would be very poor on all three institutional factors.
“There is no one quick fix to the Chinese system to make it respond better next time,” said Hung [Ho-fung Hung, a professor in political economy at Johns Hopkins University in the US]. “But if there is one single factor that could increase the government’s responsiveness to this kind of crisis, [it would be] a free press.”
‘What China’s empty new coronavirus hospitals say about its secretive system’, The Guardian, 12 Feb 2020.
4.4 Adapting Case Definitions to include potential Laboratory Exposure:
At the time of these SARS lab-escapes, the WHO and the US CDC used a definition for a ‘SARS suspected case’ that required that the case had one or more of the following exposures during the 10 days prior to onset of symptoms:
- close contact with a person who is a suspect or probable case of SARS;
- history of travel, to an area with recent local transmission of SARS;
- residing in an area with recent local transmission of SARS.
Unfortunately, an index case of a SARS Lab-escape would not meet any of these 3 criteria, potentially delaying the timely recognition and reporting of the case, as the ‘case definition’ feeds into the national early reporting systems. The ‘suspected case’ definition could thus make all the difference between an admission into a hospital with possible SARS symptoms being treated as a suspected SARS or not. This was a major problem.
In Apr 2004, in a review of the 2003 Singapore LAI published in the New England Journal of Medicine (NEJM), the authors stated that:
‘This case of laboratory-acquired SARS indicates that concern regarding the potential risk to laboratory personnel is justified, and the epidemiologic criteria in the current case definitions of the World Health Organization (WHO) and CDC may need to be amended to include laboratory exposure to SARS-CoV as a risk factor for infection.’
That proposal was a very reasonable one and was soon validated by the Beijing-Anhui outbreak and the LAIs that caused it, which were detected that very April 2004.
While that case definition discussion only applied to SARS, its validity is much larger and a laboratory exposure (direct or via contacts) should frame the definitions of suspected cases for any pathogen known to be handled there.
4.5 Managing the risk of a new biotechnology age:
We are at the cusp of a new biotechnology age. As with any new technology age, we see competition between nations driving developments and implementations that quite often are running ahead of a proper risk assessment.
It took a few unfortunate accidents for the nuclear energy sector to develop safer practices and technologies, or for some countries to simply decide that the risk/benefit did not justify the technology (see the Atomausstieg). It also took some near-misses to see a dramatic reduction in ballistic missile heads counts.
The biotechnology age is not different. Without a constant risk/benefit assessment, erring on the conservative side if necessary, we will go through the same painful learning process as we did with the nuclear age.
Easier said than done, for sure. First, we are in a context of international competition to reap the benefit of these new technologies. Secondly, contrarily to nuclear applications, the technical entry ticket cost seems much lower, while the required human ticket cost (with qualified personnel) may sometimes not be not fully paid to cut corners. To make things worse, the physical signature is much less obvious (about any nondescript urban building could contain a P2 or P3), and there is simply no international registry of P3s or even P4s.
This is not to say that all biotechnology work should be stopped. Not at all — this would be bad Luddite advice and illusory. But what it means is that the risk framework should develop in tandem with the technology, not after, unless we want to repeat the same mistakes.
References
For a quick approximate translation of pages in Chinese, please use the Google Translate extension with Chrome. It works rather well.
It is fairly common for Chinese pages to be scrubbed from the internet. If you cannot find a page given in the references, please try https://web.archive.org/. Most have been saved there. If one is still missing, please let us know (via a comment to the article). We have kept copies and can put them online.
Introduction:
[i.1] WHO SARS Risk Assessment and Preparedness Framework, Oct 2004, www.who.int/csr/resources/publications/CDS_CSR_ARO_2004_2.pdf
Singapore:
[s.1] ‘Laboratory-Acquired Severe Acute Respiratory Syndrome’, NEJM, Apr 2004, //www.nejm.org/doi/full/10.1056/NEJMoa032565
[s.2] ‘Recent Singapore SARS case a laboratory accident’, The Lancet, Nov 2003, https://doi.org/10.1016/S1473-3099(03)00815-6
[s.3] ‘Review of SARS virus leakage accidents in laboratory’, China Science News, Jul 2014, http://news.sciencenet.cn/htmlnews/2014/7/299630.shtm
[s.4] For the mention of the renovation works and mixed BSL-2/ BSL-3 activities: https://europepmc.org/article/med/14603886
[s.5] ‘International Consultant Della-Porta Challenges BSL-3 and BSL-4 Standards’ ://www.tradelineinc.com/reports/2004-9/international-consultant-della-porta-challenges-bsl-3-and-bsl-4-standards
[s.6] Details of 3 lab visits: https://www.publish.csiro.au/MA/pdf/MA08062
[s.7] 17th August: https://www.cidrap.umn.edu/news-perspective/2003/09/who-says-singapore-sars-case-not-big-concern
[s.8] Official investigation panel report — a must-read: ‘Biosafety and SARS Incident in Singapore September 2003’: https://www.nas.gov.sg/Farchivesonline/data/pdfdoc/20030923-MOH.pdf
Taiwan:
[t.1] IPMR and Taiwan bio-warfare capabilities: https://bit.ly/3ldKkaU
[t.2] For a Chinese account of the purchase of the P4 from France (codenamed “White Chicken 16”), see ‘What about biosafety! Unpopular knowledge written on National Security Day’. Saved Chinese original: https://bit.ly/3n1ZMaw, saved approximate translation: https://bit.ly/2IhqTiU. The original may be accessed (unless purged from the internet) by clicking on the first link here: https://bit.ly/3kae1Is.
We note that [t.9] confirms that all Taiwan BSL3/4 prior to 2003 were foreign-made. See also [t.18] for a confirmation of advanced French equipment.
[t.3] For Anthony Della-Porta profile as internationally renowned expert in biosecurity, biocontainment and biological safety: http://www.bio2ic.com/about/tony-della-porta
[t.4] Long hours: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7135754/
[t.5]. In a hurry: https//www.washingtonpost.com/archive/politics/2003/12/18/lab-rules-ignored-in-new-sars-case/91508154-b8dd-4ba3-a6f0-c7d002d28373/
[t.6] Many details: http://travelbanter.com/showthread.php?t=5501
[t.7] Bag: https://www.sunjournal.com/2003/12/21/officials-garbage-bag-likely-cause-infection/
[t.8] ‘He put his head inside’: the (partial) LAI database maintained by the American Biological Safety Association (ABSA) has some details otherwise unavailable, https://my.absa.org/LAI (search with keyword SARS): ‘January 7, 2004. Report on the laboratory-acquired SARS Case in Taiwan, Previously available online: During a final lab clean-up, the investigator noted “leakage from the plastic bag in the negative-pressure transport chamber”. He could not reach the spot with the attached gloves, so he wore a “normal mask” and surgical gloves to disinfect the leakage with alcohol spray. However, even with the chamber door open, he could not reach the spill, so he put his head inside the pass-through to disinfect.’
[t.9] WHO investigation and international cooperation: ‘Development of Laboratory Biosafety Management: The Taiwan Experience’ https://www.liebertpub.com/doi/pdfplus/10.1177/153567600701200104
[t.10] closure: https://www.nytimes.com/2003/09/24/world/lab-infection-blamed-for-singapore-sars-case.html
[t.11] ‘International Consultant Della-Porta Challenges BSL-3 and BSL-4 Standards’: www.tradelineinc.com/reports/2004-9/international-consultant-della-porta-challenges-bsl-3-and-bsl-4-standards.
See also the excellent https://journals.sagepub.com/doi/pdf/10.1177/153567600601100406
[t.12] Disaster: https://www.newscientist.com/article/dn4193-singapore-man-caught-sars-in-lab/
[t.13] Taiwan Epidemiology Bulletin, ‘Audit Report for Laboratories of Biosafety Level 3 and Higher in Taiwan, 2007’. https://www.cdc.gov.tw/En/File/Get/53fRU0drzfunw22tKaDplw
[t.14 ] https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(04)00911-9/fulltext
[t.15] Furmanski: https://armscontrolcenter.org/wp-content/uploads/2016/02/Escaped-Viruses-final-2-17-14-copy.pdf
[t.16] WHO post-outbreak biosafety guidelines for handling of SARS-CoV specimens and cultures. 18 Dec 2003. Accessible at: http://www.who.int/csr/sars/biosafety2003_12_18/en/
[t.17] Confirmation of leak in P4 lab, plus ‘waited 10 minutes’: https://www.publish.csiro.au/MA/pdf/MA08062
[t.18] For a mention of advanced French-made equipment: https://www.cbsnews.com/news/secret-weapon-vs-taiwans-sars/
[t.19] Travel details: https://www.cidrap.umn.edu/news-perspective/2003/12/taiwanese-sars-researcher-infected]
[t.20] Vaccine: https://english.president.gov.tw/NEWS/218
[t.21] For the full name Chan Jiacong (危家琼) and the PhD from John Hopkins: https://drive.google.com/file/d/1ve2deg-kI8sEe7Y_ds1kmMtRShBHlgN-/view
[t.22] Global Times, 10 Dec 2021, ‘Scientist in Taiwan contracts COVID-19 after P3 lab exposure, repeats SARS mistake’: https://www.globaltimes.cn/page/202112/1241199.shtml
Beijing:
The main articles of investigative journalism are:
[b.1] Caijing magazine: ‘SARS virus leak investigation of health system vulnerability’ (21 May 2004) 财经杂志:《SARS病毒泄漏调查》, http://finance.sina.com.cn/g/20040521/1530772213.shtml, also https://history.ifeng.com/c/7uAfOBruATr. Gave some details on the known infection cases at the time, some via insiders, but did not offer any explanation for the leak.
[b.2] Sichuan News Network: ‘Accountability report after the return of SARS ’ . (7 Jul 2004) 四川新闻网:《SARS重来后的问责报告焦中国疾控中心原主任》. First article to give the broad reasons for the leak based on the official report of the Ministry of Health from the 1st July (not fully made public). It does not provide details of the circumstances. http://news.sina.com.cn/o/2004-07-07/00173008395s.shtml
[b.3] Southern Weekly: ‘SARS virus leakage accident “CDC accountability incident” review’ (8 Jul 2004) 南方周末:《SARS病毒泄漏事故“疾控中心问责事件”再追踪》, https://news.sina.com.cn/c/2004-07-09/11163655901.shtml, also http://finance.sina.com.cn/careerlife/20040708/0901857988.shtml. First report giving the circumstances, especially about the sample fridge and the bad inactivation of the SARS strain.
[b.4] Xinmin Weekly: ;Virus leak: who should stand in the dock?’ (12 Jul 2004) 新民周刊:《病毒泄漏:谁该站上被告席》adds an insider report that the lab had been inspected in February and that problems had been raised, http://news.sina.com.cn/c/2004-07-12/13513683004.shtml
[b.5] China Science News: ‘Review of SARS virus leakage accident in laboratory ‘(25 Jul 2014): 中国科学报:《实验室SARS病毒泄漏事故回顾》, http://news.sciencenet.cn/htmlnews/2014/7/299630.shtm?bsh_bid=5481036177 or http://news.yanfabu.com/article-9161-1.html
[b.6] Qingdao News (27 May 204): ‘Interview with Li, the first SARS patient in Beijing this spring: no longer fear anything’ http://qingdaonews.com/content/2004-05/27/content_3175295.htm
[b.7] Beijing News, ‘The SARS accountability report announced that the time when Song was exposed to the virus is still a mystery (1 Jul 2004)’. With chain of contamination picture for 9 cases (Ren and Cui omitted): http://news.sohu.com/2004/07/01/59/news220795927.shtml
[b.8] Beijing News, ‘The SARS transmission chain is basically determined this spring (Figure)’. With important dates picture for 9 cases (Ren and Cui omitted): http://news.sohu.com/2004/07/01/59/news220795929.shtml
Official sources:
[b.9] People’s Daily Online and Jiangnan Time (20 Dec 2003), ‘The Nation’s emergency inventory of SARS laboratories’ : http://news.sina.com.cn/o/2003-12-20/10381388019s.shtml
[b.10] Beijing Evening News (19 Dec 2003), ’The nation cleans up SARS laboratories’: http://tech.sina.com.cn/other/2003-12-19/1639270834.shtml
[b.11]. For a good short biography of Prof. Hong Tao see ‘Academician Hong Tao: worked hard all his life exploring the world of viruses’ (May 2020) https://dy.163.com/article/FCSAQ4K005371QFV.html
[b.12] Beijing Evening News (19 Dec 2003): ‘The nation will be held legally responsible for the infection caused by SARS laboratories in China’. http://news.sina.com.cn/c/2003-12-19/16281383514s.shtml
The air-lock is called a ‘reflection area’ in the Chinese text, which seems to be due to the kind of reflecting light which is used there. This refers to a ‘three areas-two buffers’ concept first outlined in the 2004 ‘General Biosafety Requirements for Laboratories’: clean / potentially contaminated / contaminated areas and two air-locks in between.
[b.13] National Health and Family Planning Commission of the People’s Republic of China (13 Oct 2003), ‘Our nation SARS laboratories are safe’ http://www.nhc.gov.cn/wsb/pzcjd/200804/23067.shtml
[b.14] China Youth Daily, on Song never having entered the P3 or worked with SARS: http://news.163.com/40702/8/0Q9891R60001124T.html
[b.15] Caijing (财经, ‘financier’ in English) is a well respected magazine and is still considered as being the most independent in the whole of China — a rarity. It nevertheless ran into trouble in 2009 (see https://www.csmonitor.com/World/Global-News/2009/1110/editor-who-led-independent-journalism-in-china-resigns).
Southern Weekly (南方周末), a weekly from the staunchly innovative and argumentative southern region of China, was eventually driven to the margins by the central government (see https://foreignpolicy.com/2015/01/29/southern-weekly-china-media-censorship/).
Official conclusions:
[b51] Only extracts of the official ‘Analysis Report on the Epidemic Control of the ‘4.22’ SARS Epidemic in Beijing in 2004' (2004年北京‘4·22’SARS疫情流行控制情况分析报告) are known. The report misses the Ren and Cui cases from February. Note that the report title focuses on the April cases (‘4.22’), a possible odd justification for not mentioning the two primary February cases which explain the contamination of the lab.
[b52] The following extract includes some key tables and graph: http://www.39.net/HotSpecial/fdfy/xzbb/113011.html
[b53] Xinhuanet: Originated from Infection in the Laboratory-A Review of the SARS Epidemics in Beijing and Anhui this Spring(2004/07/01 19:12) http://news.sohu.com/2004/07/01/27/news220812709.shtml
[b54] Xinhuanet: The director of the Centre for Disease Control and Prevention responsible for the the Beijing-Anhui SARS Accidents resigned (2004/07/01 19:13), http://news.sohu.com/2004/07/01/27/news220812700.shtml
[b55] Xinhuanet: Ministry of Health announces results of investigations into the causes of SARS epidemics in Beijing and Anhui (2004/07/01 21:03), http://news.sohu.com/2004/07/01/33/news220813351.shtml
[b56] Xinhuanet: Li Liming, director of the China Centre for Disease Control and Prevention, and others resigned due to SARS in Beijing Huizhou , http://news.sohu.com/2004/07/01/24/news220802479.shtml (2004/07/01 10:21)
[b57] China Daily, ‘Officials punished for SARS virus leak’, 2 Jul 2004, www.chinadaily.com.cn/english/doc/2004-07/02/content_344755.htm
[b58] “People’s Daily”: Ministry of Health: The SARS outbreak in Beijing and Anhui this year came from laboratory infections”. 2 Jul 2004. Note how Ren and Cui cases are reported with no mention of a February infection. The original article seems to have been scrubbed out from People Daily website, but screenshots are easy to find in Chinese blogs. http://www.kunlunce.com/ssjj/guojipinglun/2020-02-18/140614.html, also archived at https://bit.ly/35wu8Mv
[b59] The WHO press release of the 2nd July 2004 can be found at https://web.archive.org/web/20040806000814/http:/www.wpro.who.int/sars/docs/update/update_07022004_revisedfinal.asp
Chinese Blog entries:
[b100] A good Chinese entry on SARS leaks: ‘Thinking: The SARS virus leak in 2004’ Automated Translation: https://bit.ly/36mIgal. Original ‘思考: 2004年SARS病毒泄露事故’, should come up as the first link in this Sogou search: https://bit.ly/2IsWRsh
[b101] Another good blog entry: ‘Investigation of SARS virus leakage in 2004’, http://www.juzizhoutou.net/tianren/lishi/2020-02-08/4868.html
[b102] For Cai Weiping’s quote see blog entry: ’17 years after SARS, The CDC is now top in the world for its ability to identify viruses’ https://bit.ly/2IsPqlr, with approximate translation: https://bit.ly/38AvY0Q
[b103] Good summary of the delays caused by the Chlamydia controversy: ‘Interview丨Hu Shuli: How was the SARS coronavirus discovered?’, https://bit.ly/3poGlKS, approximate translation: https://bit.ly/2ItZPgH
[b104] Review of “SARS Virus Leakage in China P3 Laboratory”, blog entry: https://archive.is/19WLI
[b105] 2+3 labs: ‘Did the laboratory of the Chinese Centre for Disease Control and Prevention leak the SARS virus in 2004?’, https://zhuanlan.zhihu.com/p/107322222
[b106] Another good blog entry: ‘A SARS virus leak that was about to be forgotten 16 years ago’. Original: https://bit.ly/35DWcxH, https://bit.ly/38No982 for approximate translation.
Additional Sources:
[b501] There are 4 Koch postulates in the original formulation, and 6 as modified by Rivers for viral diseases. See ‘Koch’s postulates fulfilled for SARS virus’ (May 2003) https://www.nature.com/articles/423240a
[b502] More details than published by the WHO were actually available of the site of the Chinese Consulate in Chicago on the same day: http://www.chinaconsulatechicago.org/chn/zt/sars/t91985.htm
[b503] WHO press release on 18th April: https://www.who.int/csr/don/2004_05_18a/en/
[b504] WHO press release of the 23rd April 2004, following the Chinese Ministry of Health announcement on the same day: https://www.who.int/csr/don/2004_04_23/en/
[b505] WHO Biosafety and Biosecurity in Health Laboratories — Report of a Regional Workshop, Pune, India, 8–11 July 2008. ‘SARS was acquired in the laboratory in Singapore and Taiwan in 2003 and in China in 2004. These occurred in a BSL3 laboratory in Singapore, a BSL4 in Taiwan and a BSL2 in China’. https://apps.who.int/iris/rest/bitstreams/909701/retrieve
[b506] Paper by Zhong Nanshan, ‘What we have learnt from SARS epidemics in China: ‘https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1550436/
[b507] ‘Bring critically ill patients to me’ Zhong Nanshan, https://news.cgtn.com/news/2020-09-08/Zhong-Nanshan-A-respiratory-expert-bearing-the-nation-s-trust-TBcBq2soqk/index.html
[b508] Science Mag: ‘SARS Crisis Topples China Lab Chief’, Jul 2004: https://www.sciencemag.org/news/2004/07/sars-crisis-topples-china-lab-chief
[b509] WHO press release of the 16th Apr 2003 confirming that SARS is caused by a coronavirus: https://www.who.int/csr/don/2003_04_16/en/
[b510] WHO press release on 3rd Apr 2003 stating that ‘SARS, [..] is thought to be caused by a new virus in the coronavirus family’: https://www.who.int/csr/don/2003_04_03a/en/
[b511] All WHO SARS ‘Disease outbreak news’: https://www.who.int/csr/don/archive/disease/severe_acute_respiratory_syndrome/en/
[b512] WHO: ‘China confirms SARS infection in another previously reported case; summary of cases to date — Update 5’, 30 Apr 2004.www.who.int/csr/don/2004_04_30/en/
[b513] Bulletin of the World Health Organization, June 2004. Note the absence of mention of the two primary February cases (but an indirect reference to work on SARS in February and March). Also note: ‘Dr Merianos, who recently visited the Institute for Virology in Beijing, could not say whether laboratories here or in other Chinese research facilities adhered to biosafety level 3.’ https://www.who.int/bulletin/volumes/82/6/en/news.pdf
[b514] Interestingly, a paper by some Chinese scientists confirmed the count of 11 cases: Biomed Environ Sci, Dec 2006, a : ‘Severe acute respiratory syndrome — retrospect and lessons of 2004 outbreak in China’. Requests to get access to the full paper were not answered by the authors. https://pubmed.ncbi.nlm.nih.gov/17319269/.
[b515] WHO, 29 Apr 2004: ‘China confirms SARS infection in two previously reported cases — update 4’, https://www.who.int/csr/don/2004_04_29/en/
[b516] The-scientist.com, ‘SARS escaped Beijing lab twice’, Apr 2004 www.the-scientist.com/news-analysis/sars-escaped-beijing-lab-twice-50137
[b517] Article about a mobile P3 lab set in the old Institute building, ‘Between “yin and yang”, the reporter directly hit the laboratory test of the Institute for Disease Control and Virus Diseases’, Jun 2020 https://www.sohu.com/a/404191251_206055
[b518] ‘Additional patients in China under investigation for SARS; WHO team travels to Beijing — update 2’, 26th Apr 2004, https://www.who.int/csr/don/2004_04_26/en/
[b519] ‘Surveillance and control of infectious atypical pneumonia in the interior of the country from January to May 2004’, 28 Jun 2004, http://www.nhc.gov.cn/bgt/pw10405/200406/521b9e32e20247619421253c0d7a863b.shtml
[b520] WHO Situation Updates — SARS, https://www.who.int/csr/sars/archive/en/
On the chlamydia controversy
[b200] For an excellent recap of the events: ‘SARS virus hits China’s scientific research system’, Jun 2006, https://news.sina.com.cn/c/2003-06-11/01461156694.shtml, also through http://blog.sina.com.cn/s/blog_8d622a9e0100xgqb.html
[b201] A good recap of the Chlamydia hypothesis is available in ‘Hong Tao Chinas National CDC and the Chlamydia Hypothesis’, Flanders Health Blog, https://www.flandershealth.us/sars-outbreak/hong-tao-chinas-national-cdc-and-the-chlamydia-hypothesis.html
[b203] For some important quotes from Hong Tao see The Bund Magazine (上海外滩画报) (2003 Apr 24) https://news.sina.com.cn/c/2003-04-24/08441013638.shtml
[b204] For the paper by Hong Tao linking SARS to Chlamydia: https://mall.cnki.net/magazine/Article/ZHYX200308004.htm, Aug 2008
[b205] Mistaken Identity of Germ Culprit Cost the Chinese Time and Prestige, WSJ, Jun 2004: https://www.wsj.com/articles/SB105467896681095400https://www.wsj.com/articles/SB105467896681095400
[b206] “Virologist Hong Tao, [ — ] said they had detected traces of coronavirus from three tissue samples. However, the team has not conducted follow-up research because it does not consider it a research focus.” http://www.china.org.cn/english/features/SARS/61091.htm
[b207] For the main paper by Hong Tao setting the case: ‘Chlamydia-like and coronavirus-like agents found in dead cases of atypical pneumonia by electron microscopy’ (1st Apr 2003): https://europepmc.org/article/med/12887816
“CONCLUSION: Since the novel Chlamydia-like agent was found co-existing with a coronavirus-like agent in the dead cases of SARS, it looks most likely that both the agents play some roles in the disease. At the present time, however, one can hardly determine how these agents interact with each other synergistically, or one follows another — this needs further study”
On nepotism:
[b301] Yi Ming, The problems of Chinese academia and their way out, Jul 2004 (亦明, 中国学术界的问题及其出路), http://bbs.tianya.cn/post-free-179032-1.shtml. Also, https://drive.google.com/file/d/1XyWj30Pcpp3Ru1o5m1jML6L6wVWc5dmz/view?usp=sharing for translation and https://drive.google.com/file/d/1vQ-yGL47x6VOBLiWgee5zydfog1Hzv_h/view?usp=sharing for a copy of the original.
Note this excellent passage:
There was only one predecessor of these three laboratories, the laboratory of virus morphology and viral diarrhoea lab. In other words, yet another academician will not only create two more laboratories, but one of them is an “academician” laboratory showing that he does not even know what he is doing in it.
“It is no wonder that these people not only fail to share the worries and alleviate problems for the country, but also add chaos to the country. I have seen with my own eyes that a so-called senior academician controls a laboratory worth millions of dollars, but there is no one inside. Just because he is an academician, others dare not speak up against him. It was also this academician, shaking a few pieces of paper with the genetic sequence that had been stored in GenBank long ago, declaring that it was a scientific secret.
In fact, those “authorities” who have long lost their creative ability (in fact, many of them have never had any creative ability), the most fearful thing is to be looked down upon by others, afraid of being overtaken by others, so they cultivate a direct line in academia, a gang, and the goal of competition between gangs is not who has a new academic achievements, but who wins the prize.
The director of the laboratory that released the SARS virus again this year, the viral diarrhoea laboratory of the Institute for Viral Disease Control and Prevention, is the proud student of Academician Hong Tao. It’s really “Where there is a teacher, there is a disciple” (有其师必有其徒)! The academician system has also set an example for new academics in China: Those who work hard and do research are idiots, and those who can get on with nothing, or less, are “brilliant people with leadership skills.”
[b302] For a good critic of the academic system in China, see ‘The disease of science lies in culture’, a discussion with Rao Yi, trained in the US (UCSF, Harvard) and known for his fight against corruption in the academic system. https://www.sohu.com/a/48091593_183834
[b303] Wang Jiangwei’s CV: https://bit.ly/2IKlIZ6, archived here: https://bit.ly/3q4HHe9 (original), https://bit.ly/3fMhg8A (translation)
[b304] ‘Introduction to Laboratory Biosafety -Wang Jianwei’, https://wenku.baidu.com/view/1caa674169eae009581beca8.html, archived at https://bit.ly/38KnWSW
[b305] For Dong Xiaoping’s role on WHO-China Coronavirus Disease Joint Expert Investigation Group: see https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19---final-report-1100hr-28feb2020-11mar-update.pdf?sfvrsn=1a13fda0_2
[b306] Hong Tao’s medal for his contribution to a healthy China and epidemics prevention: https://bit.ly/3o16OwU (original), https://bit.ly/3q80Hsf (translation)